Revolutionary Tiny Pacemaker Offers Non-Surgical Solution for Pediatric Heart Patients
April 2, 2025
Despite its tiny size of 1.8 mm wide and 3.5 mm long, the pacemaker delivers the same level of stimulation as a full-sized unit, making it suitable for patients requiring temporary pacing.
Controlled by a light-sensitive switch, the device can be activated using infrared light emitted from a soft, wireless wearable patch that monitors heart rate and adjusts pacing accordingly.
The device has shown promising results in both animal models and human hearts from deceased donors, paving the way for future human trials.
Key researchers, including Professors John Rogers and Igor Efimov, emphasize the critical need for miniaturized temporary pacemakers in pediatric surgeries, highlighting the potential to minimize infection risks and complications.
Future advancements may allow for the deployment of multiple pacemakers across the heart, enhancing treatment options for complex conditions like arrhythmias.
The research team has established a company, NuSera Biosystems, to further develop the device and seek FDA approval for clinical trials.
This development builds upon previous work on a biodegradable, quarter-sized pacemaker, addressing limitations related to receiver antenna size.
Experts in the field have praised this development as a significant breakthrough in miniaturization, with potential applications extending beyond cardiac care to areas like nerve healing and pain management.
Researchers have developed a groundbreaking temporary pacemaker that is smaller than a grain of rice and can be injected into the body, eliminating the need for surgical installation and removal.
This innovative device is designed primarily for pediatric care, as approximately one percent of children are born with congenital heart defects, making it particularly beneficial for newborns during critical recovery periods after heart surgery.
The pacemaker operates without external power, utilizing a galvanic cell powered by the body's biofluids, which simplifies the implantation process and reduces patient trauma.
The study detailing this breakthrough was published in the journal Nature on April 2, 2025, and emphasizes the importance of this innovation for improving patient comfort and safety.
Summary based on 11 sources
Get a daily email with more Science stories
Sources

Mashable • Apr 3, 2025
Tiny pacemaker smaller than a rice grain designed for newborn babies
Nature • Apr 2, 2025
World’s tiniest pacemaker could revolutionize heart surgery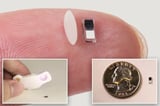
New York Post • Apr 3, 2025
'World's smallest pacemaker' is implanted with a syringe and dissolves when it's no longer needed