UK Authorizes Revolutionary Gene Therapy, Casgevy: A Potential Cure for Sickle Cell Disease
November 16, 2023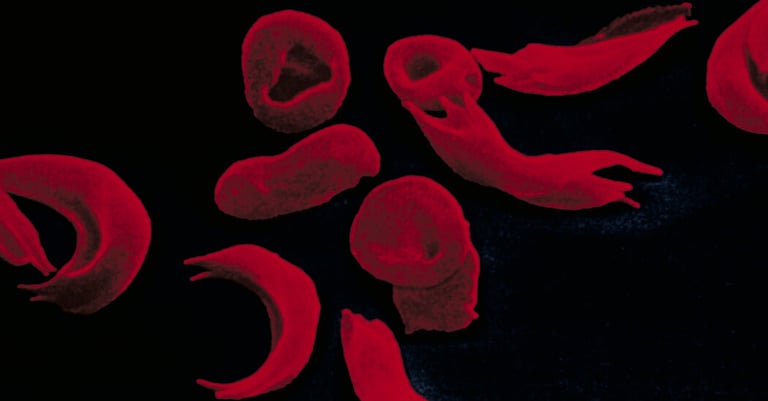
The MHRA, the UK's medicines regulator, has authorized the use of Casgevy, a gene therapy for sickle cell disease and beta thalassemia.
Casgevy uses the gene-editing tool Crispr to modify the patient's own stem cells, eliminating the need for immune compatibility and immunosuppression.
Clinical trials have shown promising results, with patients seeing healthy hemoglobin production restored and a decrease in symptoms.
Regulators in other countries, including the US, are expected to approve the therapy.
Concerns persist about the cost and accessibility of the treatment, especially in Africa where most sickle cell disease cases are found.
The therapy is also currently under review in the US and the European Union.
Summary based on 0 sources